Abstract
Introduction: Primary immune thrombocytopenia (ITP) is an immune-mediated disorder characterized by isolated thrombocytopenia and risk of hemorrhage. Impaired megakaryocyte maturation and insufficient platelet formation, induced by platelet-specific autoantibodies or ITP plasma, play vital roles in its pathogenesis. Although thrombopoietin receptor agonists (TPO-RA) is recommended the first choice among second-line treatment of ITP, clinical data have indicated that nearly half of ITP patients failed or relapsed during eltrombopag maintenance, and increasing drug dose cannot reverse therapeutic response. Diacerein, hydrolyzed into its active metabolite rhein after oral administration, is a non-steroidal anti-inflammatory drug used in osteoarthritis. Our team first observed that 3 ITP patients with low platelet levels (30-50 ×109/L) during eltrombopag treatment, achieved significant platelet response (≥100×109/L) after taking diacerein concomitantly due to the complaint of arthralgia. These data suggested that diacerein may have sensitization effect on TPO-RA, and this novel combination is expected to boost thrombopoiesis in ITP. Thus, in vitro and in vivo experiments were performed to explore the role of TPO-RA plus rhein in megakaryocyte growth and platelet formation in ITP.
Methods: Twenty-eight eltrombopag-inefficient or relapsed ITP patients and 15 healthy volunteers were included in this study. We sorted CD34+ cells from healthy umbilical cord blood mononuclear cells, then cultured them in SFEM medium containing thrombopoietin, interleukin-3, stem cell factor and 10% serum from either ITP patients or healthy donors. Different doses of eltrombopag combined with rhein (0-100 μM) were added on the fourth day, and cell suspension was collected on the 14th day. Flow cytometry, RNA sequencing, proteomic sequencing and cytokine assay were performed to evaluate the overlaying effect and underlying mechanisms of rhein on eltrombopag. In addition, active ITP murine models were established to evaluate the therapeutic effects of romiplostim plus rhein in vivo.
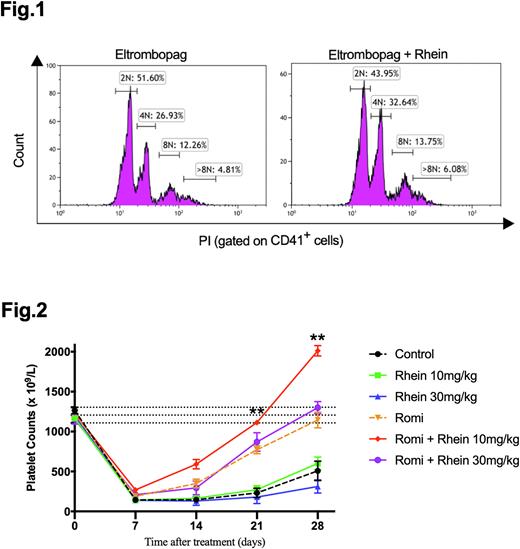
Results: Compared with eltrombopag, eltrombopag combined with low-dose rhein significantly increased the proportion of CD41+, CD41+CD42+, CD41+CD61+ megakaryocytes in vitro. We further found that eltrombopag plus low-dose rhein significantly promoted polyploid maturation and apoptosis of CD41+ megakaryocytes, as well as platelet production in vitro (Fig 1). The ITP mice exhibited profound thrombocytopenia, and their platelet counts decreased significantly by the second week after irradiation. Our data showed that romiplostim plus low-dose rhein significantly alleviated thrombocytopenia than romiplostim monotherapy on day 21 and day 28 (Fig 2). Mice receiving different doses of rhein monotherapy were comparable in platelet counts with control group. Flow cytometry analysis suggested that the combination therapy significantly increased megakaryocyte percentage and polyploidization than romiplostim in ITP mice. Subsequently, RNA sequencing and proteomic analysis revealed that the combination of eltrombopag and low-dose rhein promoted megakaryocyte growth and platelet formation, decreased proinflammatory cytokines, and upregulated phosphorylated PI3K in vitro. Cytokine analysis indicated that eltrombopag plus low-dose rhein significantly increased supernatant level of TGF-β, and reduced IFN-α, IFN-γ, IL-12p70, IL-17A and IL-18 than eltrombopag. Furthermore, it was confirmed that the addition of rhein to TPO-RA significantly promoted megakaryocyte PI3K phosphorylation in vitro and in vivo. PI3K inhibition offset the enhanced effect of eltrombopag plus low-dose rhein over eltrombopag in megakaryocyte maturation and platelet formation.
Conclusions: In sum, our data reveal a sensitizing role of rhein to TPO-RA in megakaryocyte polyploidization and platelet formation in ITP, and this novel combination therapy warrants further investigation as an optimized strategy for ITP patients.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal